
(A) catalyzes formation of sugar-phosphate bonds in adjacent Okazaki fragments (B) produces breaks in the DNA molecules and then rejoins the strands to prevent supercoiling (C) joins successive nucleotides to a growing polynucleotide strand. Our results show that the interaction between PCNA and DNA ligase I has a key role in long-patch BER and provide the first evidence for the biological significance of this repair mechanism. Study with Quizlet and memorize flashcards containing terms like In the following questions, match the component of DNA replication with its function. Extracts from 46BR.1G1 cells were defective in long-patch, but not short-patch, base-excision repair (BER). 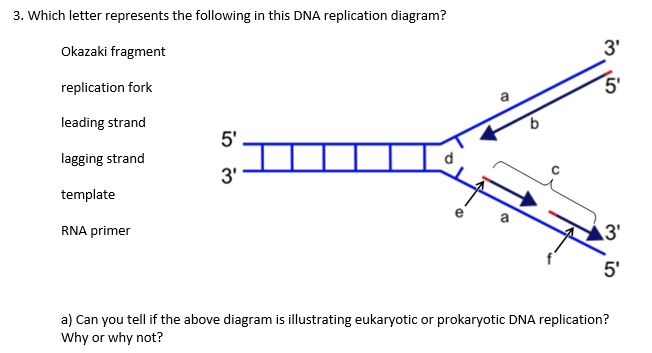
A functional PCNA-binding site was also required for the ligase to complement hypersensitivity of the DNA ligase I mutant cell line 46BR.1G1 to monofunctional alkylating agents, indicating that a cytotoxic lesion is repaired by a PCNA-dependent DNA repair pathway. Okazaki fragments also contain RNA primers, which first need to be removed by Flap endonuclease I, and replaced with new DNA added by DNA Polymerase. In short, the RNA primers are first removed by Flap endonuclease I and replaced by DNA Polymerase, after which these fragments are joined by DNA ligase I. This is why joining Okazaki fragments together is a bit longer process. Thus, the interaction between PCNA and DNA ligase I is not only critical for the subnuclear targeting of the ligase, but also for coordination of the molecular transactions that occur during lagging-strand synthesis. Okazaki fragments also contain RNA primers, which need to be replaced by DNA fragments. DNA polymerase Primase Helicase DNA ligase DNA ligase is an enzyme used to facilitate the joining of the DNA strands together by catalyzing the formation of. In contrast, the loss of PCNA binding severely compromised the ability of DNA ligase I to join Okazaki fragments. There is compelling evidence that DNA ligase I is predominantly responsible for joining Okazaki fragments generated by discontinuous DNA synthesis on the. The RNA nucleotides from the short RNA primers must be removed and replaced by DNA nucleotides, which are then joined by the DNA ligase enzyme. The synthesis of the lagging strand is discontinuous since the newly formed strand is disjointed. Inactivation of the PCNA-binding site of DNA ligase I had no effect on its catalytic activity or its interaction with DNA polymerase beta. These short fragments are known as the Okazaki fragments. The surprising ability of mouse lig1 null cells to proliferate suggests that another DNA ligase, most likely DNA ligase III, is able to join Okazaki. Here we examine the biological significance of this interaction. DNA, removal of the RNA primer, its replacement by a stretch of DNA, and the covalent linking of adjacent Okazaki. For example, DNA ligase I-deficient cells exhibit a marked defect in Okazaki fragment joining, and DNA ligase I physically and functionally interacts with the replication proteins, PCNA and RFC. 
DNA ligase I belongs to a family of proteins that bind to proliferating cell nuclear antigen (PCNA) via a conserved 8-amino-acid motif.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
